How Many Atoms in a Mole of Sodium
Substitute for for and for in equation 1. 13 What is the equivalent weight of NaHCO3.

Pin By Ncert Solutions On Aplus Topper Particles Carbon 12 Water Molecule
The gram atomic mass of sodium Na is 23 g.

. 11 How do you calculate moles of NaHCO3. 5 g of N a. 4moles of Na 6022 x 10 23 atoms1 mole of Na240 x 10 24.
How many atoms are in a 122 mole of sodium. Then multiply the number of moles of Na by the conversion factor 602214179 10 23 atoms Na 1 mol Na with 602214179 10 23 atoms being the number of atoms in one mole of Na Avogadros constant which then allows the cancelation of moles leaving the number of atoms of Na. 3 mol Na 60221023 atoms Na1mol Na 18071024 atoms Na.
One mole of a substance is defined by Avogadro as consisting of 6022 x 1023 atoms. Write the equalities for going from moles of. 14 What is the mass of 100 mole of aspartame.
View solution Which has more number of atoms. The molar mass of Na is 22989 gmol. To calculate mass multiply given moles by molar mass Na.
Sodium atomic number is 11. On putting the respective values in formula 1 we get. Using dimensional analysis and avogadros number you can get the number of atoms in Na.
The number of atoms per mole of any element or the number of molecules per mole of any compound is 60223 x 1023 atoms. View solution Calculate. N 23 23.
Want to see the full answer. One mole of atoms is 60221023 atoms. 1 mole of atoms is equal to 1 avagadro number of atomsie 60221023.
12 How many moles of sodium hydrogen carbonate NaHCO3 are in a 508 g of NaHCO3. Number of moles G i v e n m a s s G r a m a t o m i c m a s s. Atoms Na 122 mole Na 602 x 1023 atoms Na----- x -----1 1 mole Na 734x1023 atoms Na.
1 mole of S atom 602 x 10 23 atoms of S. Thus 23 g of sodium contains one mole of sodium atoms. The number of moles of sodium atom in 23g of sodium is 1 mole.
How many atoms are in 5 moles of sodium. Check out a sample QA here. This is Avogadros number.
3 mol Na 22989 g Na1mol Na 68967g Na. To calculate atoms from moles multiply given moles by 60221023 atomsmol. 7 What is the molar mass of na2so3.
Therefore by multiplication 6. 1 molecule H O2 atoms H 1 mole of H2O2 moles of H 1 molecule H0 1 atom O 1 mole H 01 mole o Just like how if it takes 3 eggs to make 1 cake it would take 3 dozen eggs to make 1 dozen cakes. Students whove seen this question also like.
To find the number of atoms in the given moles of the element follow the following steps. In one mole of sodium phosphate three moles of sodium atoms one mole of phosphorous atoms and four moles of oxygen atoms are present. How many moles of sodium are in Na2CO3but in ONE mole quantity of sodium carbonate there are TWO moles of sodium atoms with a mass of 46g one mole of carbon atoms with a mass of 12g and THREE moles of oxygen atoms with a mass of of 48g.
Check out a sample QA here. Mass number is 2298. 1 Mole 60221415E23 Atom.
To calculate the number of atoms in two moles of sodium use dimensional analysis. 20 moles Na x 60221023g1mol1201024 atoms of Na. Want to see the full answer.
Additionally how many atoms are in a mole of sodium. 15 How do you find the. 10 How many moles of water h2o are present in 750 g h2o.
Then multiply the number of moles of Na by the conversion factor 6022141791023 atoms Na 1 mol Na with 6022141791023 atoms being the number of atoms in one mole of Na Avogadros constant which then allows the cancelation of moles leaving the number of atoms of Na. 100g of sodium or 100g of iron. Therefore one mole of sodium 2298 grams of sodium.
How many atoms are in a gram. How many atoms are contained in a mole of acetic acid. N a 2 3 u S 3 2 u O 1 6 u N A.
Where Atom Number of atoms. Use the avogadro number to convert the moles to atomsWe know that 1 mole of any atom compoundformula unit has 602 x 10 23 atomcompound or formula unit In the problem given here there are there are 2 moles of S atom. 6 How many so32 ions are contained in 196 mg of na2so3 the molar mass of na2so3 is 12605 g mol.
10 moles of sodium 102298 2298 grams of sodium. 9 How many protons are in 1 mole of copper atoms. The definition of Avogadros number of 6022 1023mole is the number of atoms or molecules per one gram atomic weight.
Therefore the molar mass of sodium phosphate is 16397 gmol. We know that 6023 10 23 atoms are present in one mole of any substance. Atomic mass of Na 23u Fe 56u.
Therefore 2298 grams of sodium consists of 1 mole of sodium atoms. The number of atoms in 1 1. How many atoms are in a 122 mole of sodium.
N 1 m o l e.

Infographic Database Infographic Examples For Your Inspiration Chemistry Classroom Chemistry Lessons Teaching Chemistry
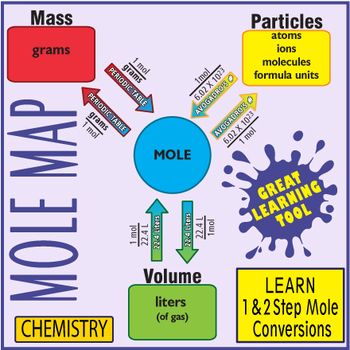
My Chemistry Students Find Using This Map The Easiest Way To Quickly Master 1 And 2 Step Mole Conversio Teaching Chemistry Chemistry Education Chemistry Basics

1 Mole Number Of Atoms In 12 G Of Carbon Chemistry Classroom Teaching Chemistry Chemistry Lessons
Comments
Post a Comment